4Front ProAudio VST/VSTi



| 4FRONT TruePianos TruePianos is a new virtual piano VSTi based on a combination of physical modeling, synthesis and sampler techniques. Using a combination of the best what these individual technologies have to offer, TruePianos provides great playability by allowing itself to be easily adjusted to the combination of your unique playing style and the specific characteristics of your MIDI keyboard, instead of the other way around. It doesn't attempt to meticulously simulate existing pianos but instead provides realistic and expressive range of piano modules, each with individual dry sounding presets that you 'just play'. |
||
|
The blue precipitate dissolves, forming a bright blue (cornflower blue) transparent solution. This indicates the presence of multiple hydroxyl ( −OHnegative cap O cap H ) groups on adjacent carbon atoms. Step C: Identifying Aldehydes or Glucose Below is a structured report (essay-style) for this practical work. 1. Define the Objective Both aldehydes and glucose (which contains an aldehyde group) react with upon heating. Add and heat the mixture gently. The primary goal of this experiment is to use qualitative chemical reactions to identify unknown organic substances in test tubes, such as . 2. Prepare Reagents and Equipment Polyhydric alcohols react with copper(II) hydroxide at room temperature. Add freshly prepared to the test tube. Carboxylic acids can be identified by their acidity or their reaction with carbonates. Add a drop of litmus or a small amount of Na2CO3cap N a sub 2 cap C cap O sub 3 to the sample. Observation: Litmus turns red; Na2CO3cap N a sub 2 cap C cap O sub 3 produces effervescence (bubbles of CO2cap C cap O sub 2 Equation: |
||
| 4FRONT BASS MODULE (VSTi) The bass module is a morph between sample playback and a synthesizer. Original string excitation is sampled, and then sound is modeled. Playing chords will add extra fat rumble, like when playing a real bass guitar. Bass guitar range was artifically extended up and down, so that higher notes and parts can be played as well. |
||
|
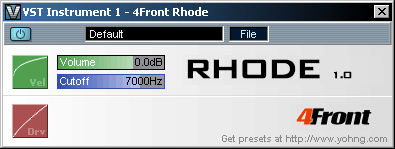
4FRONT RHODE MODULE (VSTi)
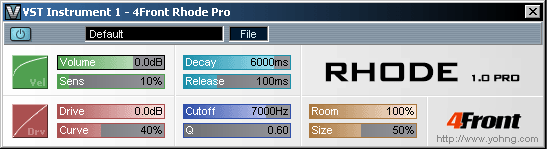
A vintage Rhodes/Wurly typed piano module with gentle overdrive. This module reproduces the classic sound similar to Rhodes/Wurlitzer pianos. Module is not sample based, and the sound is generated on-the-fly, therefore there are no sampling layer switches - vintage rhodes overdrive is smooth, clean and continuous. Difference between Basic (free) and Pro (commercial) modules To get a common idea of what exactly is different, check the screenshots below: Basic version:
As you can see, basic version is generally lacking envelope, sensitivity/drive and room controls, that Pro version has. If you like to get this kind of controlling over 4Front Rhode sound, you may want to get Pro version. MP3 Demos |
||
| 4FRONT E-PIANO MODULE (VSTi) E-Piano module with a rich, warm and silky sound. It is quite unique by its character. The module is made with a hybrid technology, which involves sample playback and synthesizing within one case. For its quality, module has reasonable small size. |
||
Khimiia 10 Klass Rudzitis 2 Prakticheskaia Rabota OfficialThe blue precipitate dissolves, forming a bright blue (cornflower blue) transparent solution. This indicates the presence of multiple hydroxyl ( −OHnegative cap O cap H ) groups on adjacent carbon atoms. Step C: Identifying Aldehydes or Glucose khimiia 10 klass rudzitis 2 prakticheskaia rabota Below is a structured report (essay-style) for this practical work. 1. Define the Objective The blue precipitate dissolves, forming a bright blue Both aldehydes and glucose (which contains an aldehyde group) react with upon heating. Add and heat the mixture gently. The primary goal of this experiment is to The primary goal of this experiment is to use qualitative chemical reactions to identify unknown organic substances in test tubes, such as . 2. Prepare Reagents and Equipment Polyhydric alcohols react with copper(II) hydroxide at room temperature. Add freshly prepared to the test tube. Carboxylic acids can be identified by their acidity or their reaction with carbonates. Add a drop of litmus or a small amount of Na2CO3cap N a sub 2 cap C cap O sub 3 to the sample. Observation: Litmus turns red; Na2CO3cap N a sub 2 cap C cap O sub 3 produces effervescence (bubbles of CO2cap C cap O sub 2 Equation: |
||
|
4FRONT AUXITER (VST/DX)
This is a high frequency refresher exciter-type plugin. It brings clarity to the high end by regenerating high frequency harmonics. Primary design goals: - Vocal enhancer - Per/track enhancer - Master track restoration/refreshment - Old tracks restoration Usage: First try to figure right Frequency slider value for your track, then control the Harmonic, Drive and Mix levels to get the best enhancement. 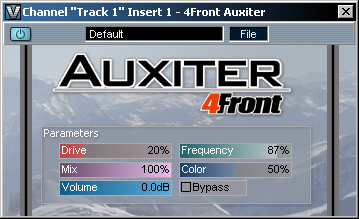 |
||
| 4FRONT SAND BRUSH (VST/DX) The plugin completely regenerates high frequencies, basing on the existing partials. The plugin can be used to apply on hihats and other percussive sounds with high frequency content, and also can be used on other tracks to add "sand". Depending on the settings, plugin can be also used to emulate the vintage vinyl sound. Primary design goals: - Sand for vocal and solo tracks - Bring life back to sampled strings - Track restoration (including MP3) - Vintage effect (to create vintage sound) The plugin contains multiple presets for various track types. 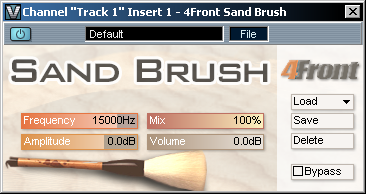 |
||
| 4FRONT CONTOUR MAX (VST/DX) The plugin is designed to change the basic frequency contour of a sound track, or the overall frequency balance of a master. Primary design goals: - Master processing - Per-track processing for balancing low and high end - Track restoration The plugin contains multiple presets for various track types.  |